Clinical Trials - Grey Literature in the Health Sciences - Research Guides at University of Alabama - Birmingham

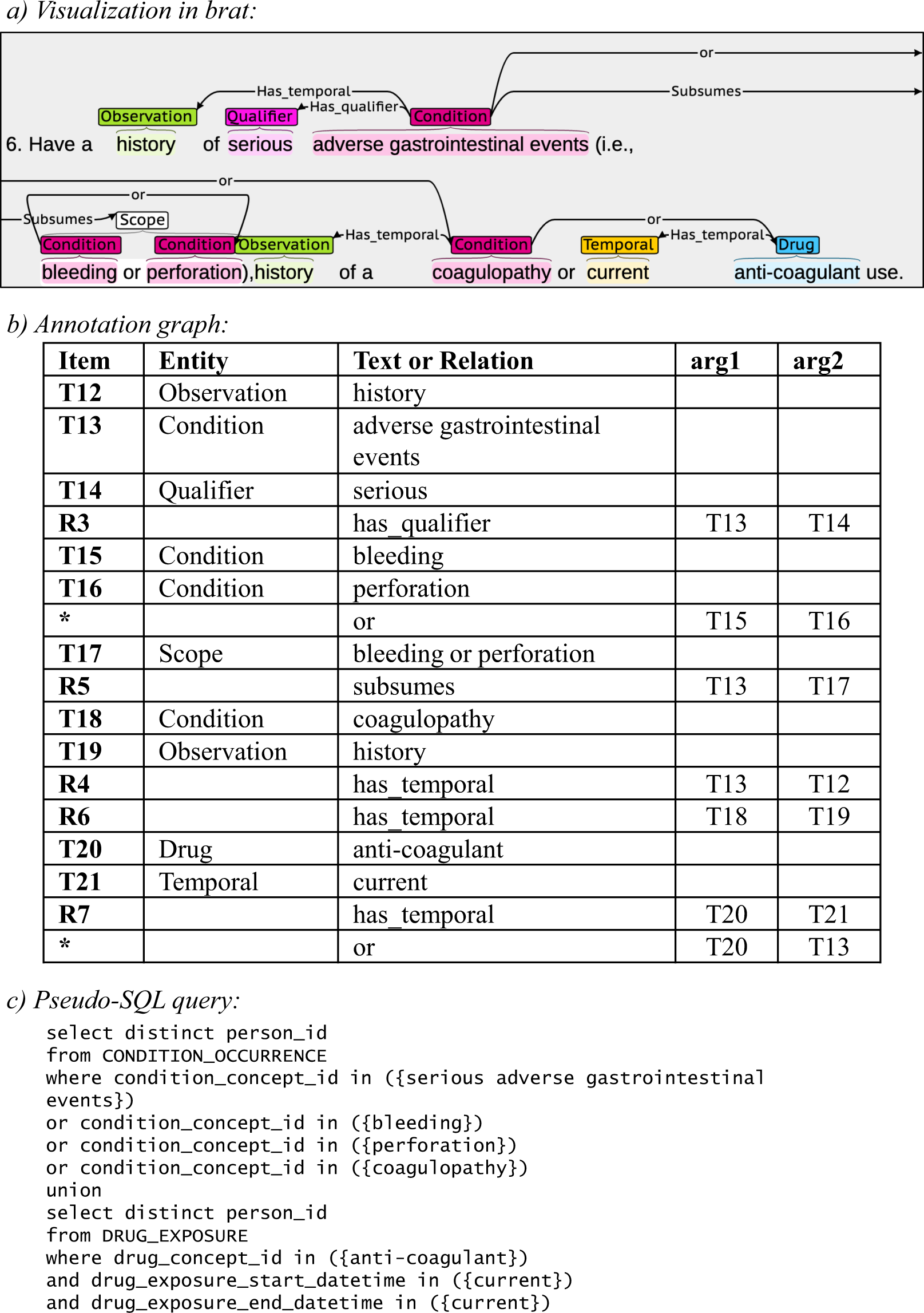
A schematic representation of the database for Aggregate Analysis of... | Download Scientific Diagram

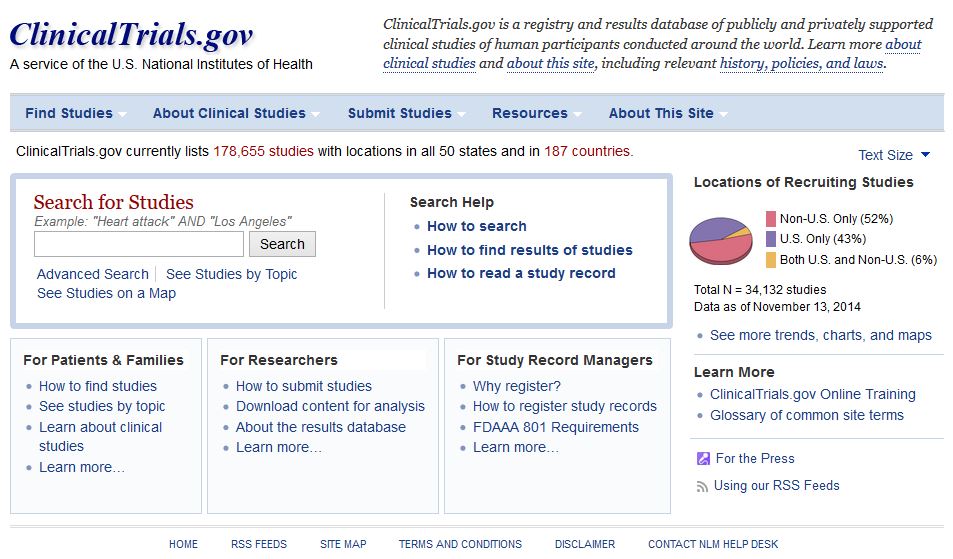
Celebrating 20 Years of ClinicalTrials.gov and Looking to the Future – NLM Musings from the Mezzanine

Compliance with legal requirement to report clinical trial results on ClinicalTrials.gov: a cohort study - The Lancet

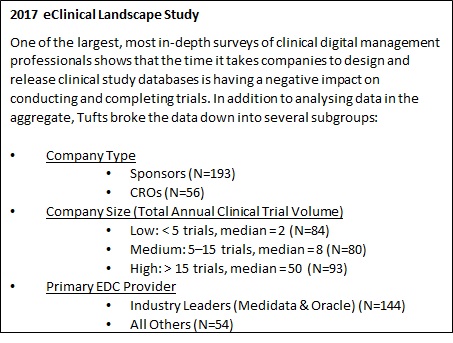
Statistical controversies in clinical research: limitations of open-label studies assessing antiangiogenic therapies with regard to evaluation of vascular adverse drug events—a meta-analysis - Annals of Oncology

Result dissemination from clinical trials conducted at German university medical centers was delayed and incomplete - ScienceDirect

Compliance with requirement to report results on the EU Clinical Trials Register: cohort study and web resource | The BMJ

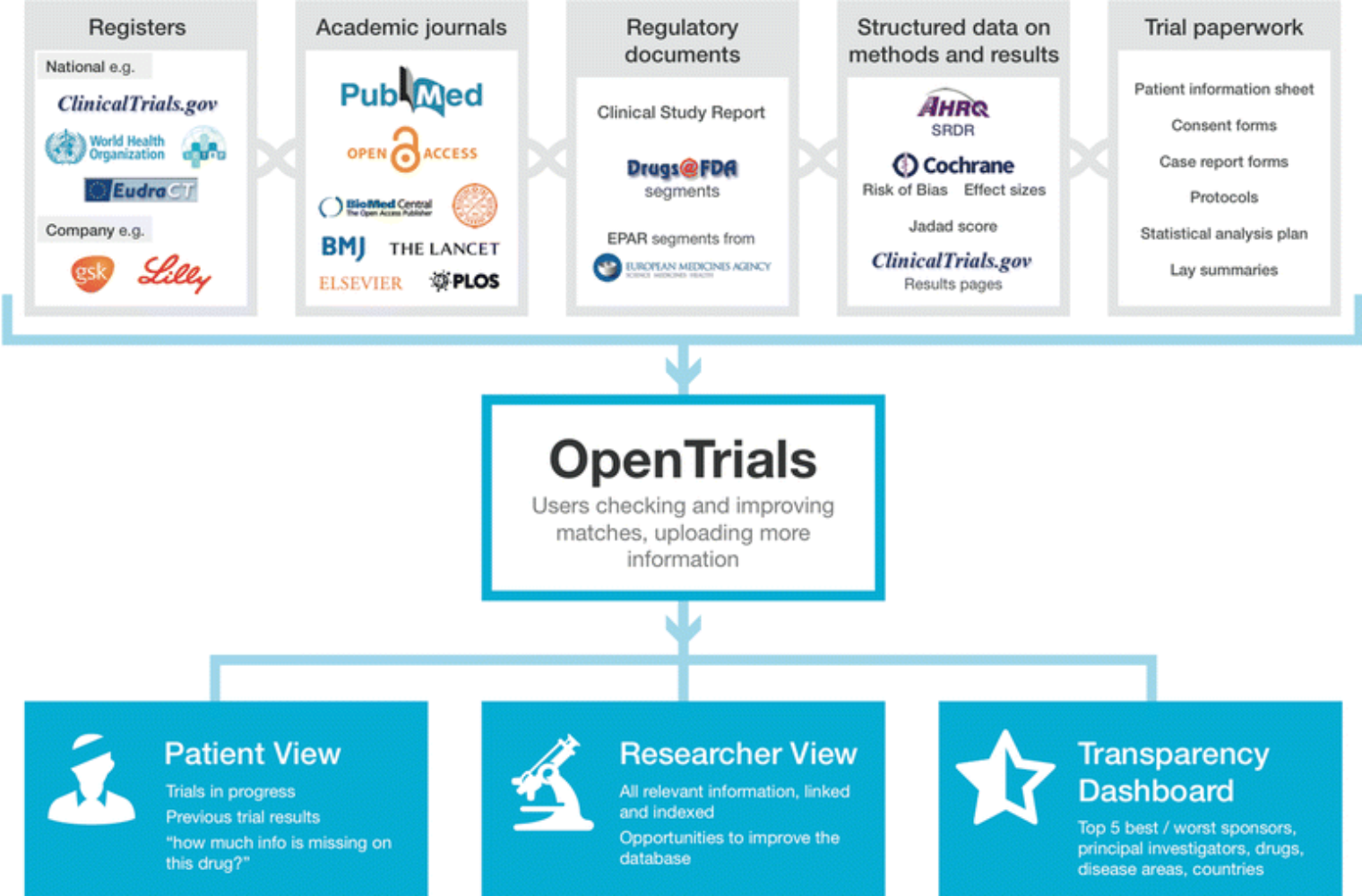
Clinical Trial Transparency: Ensuring Compliance with the New FDA Guidelines | BrackenData Clinical Trial Intelligence