
Study on the structural degradation of (a−d) FL-BP acetone and (e−h)... | Download Scientific Diagram
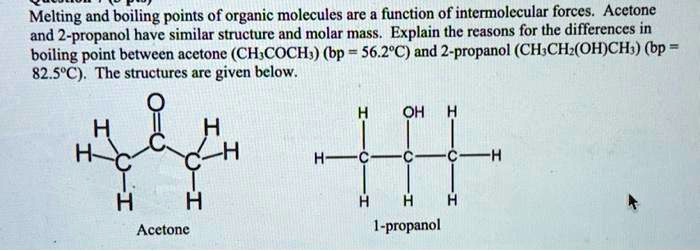
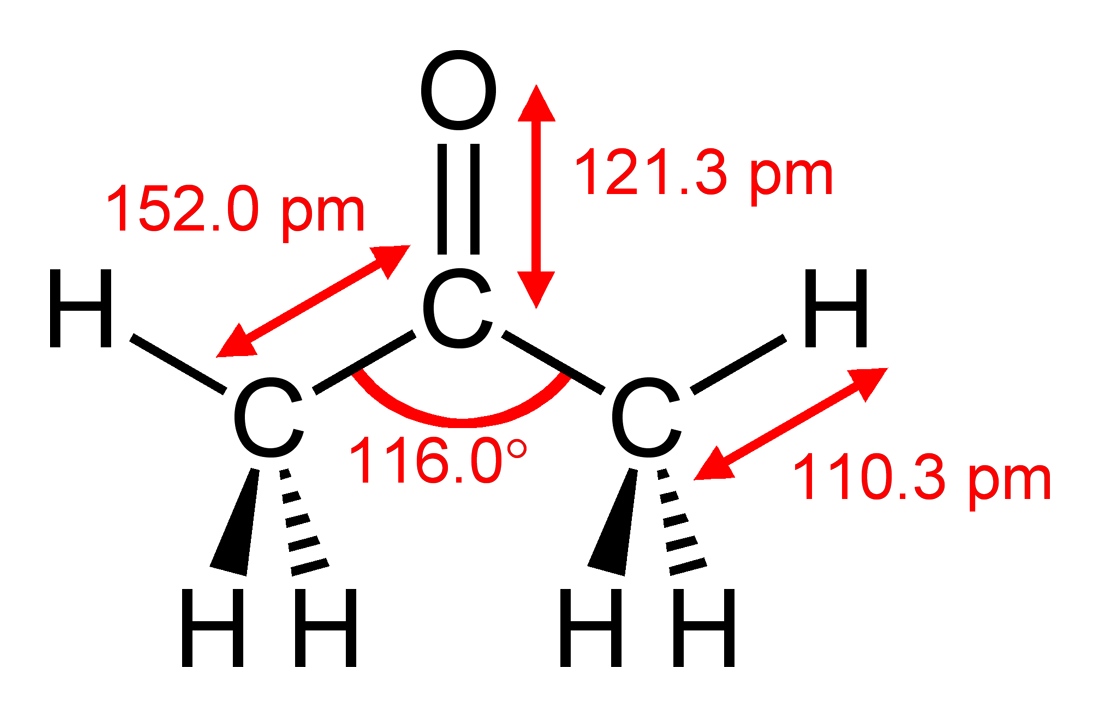
SOLVED: Melting and boiling points of organic molecules are function of intermolecular forces: Acetone and 2-propanol have similar structure and molar mass. Explain the reasons for the differences in boiling - point

Resistance change of MWCNT BP immersed in acetone (a), ethanol (b) and... | Download Scientific Diagram

Resistance change of MWCNT BP immersed in acetone (a), ethanol (b) and... | Download Scientific Diagram

SOLVED: (bp=5o"C): The Acetone (bp-56"C) and n-hexane (bp-70*C) form a binary minimum boiling azeotropei 'distilled composition of the azeotrope is 60% acetone and 40% n-hexane: Suppose that you fractioeiallyadi 100.0 mL sample

a) STEM image of FL-BP acetone sheets. (b) STEM image of a selected... | Download Scientific Diagram

Acetone BP, Propanone, CAS No 67-64-1, Dimethyl Ketone, Ketone propane, Pyroacetic spirit in Mumbai , Polypharm Private Limited | ID: 4231576297

GREEN - Research Tip of the Month: Recycling Acetone Most organic chemists doing synthesis in the lab have to clean their glassware to remove the current reaction to avoid contamination of future









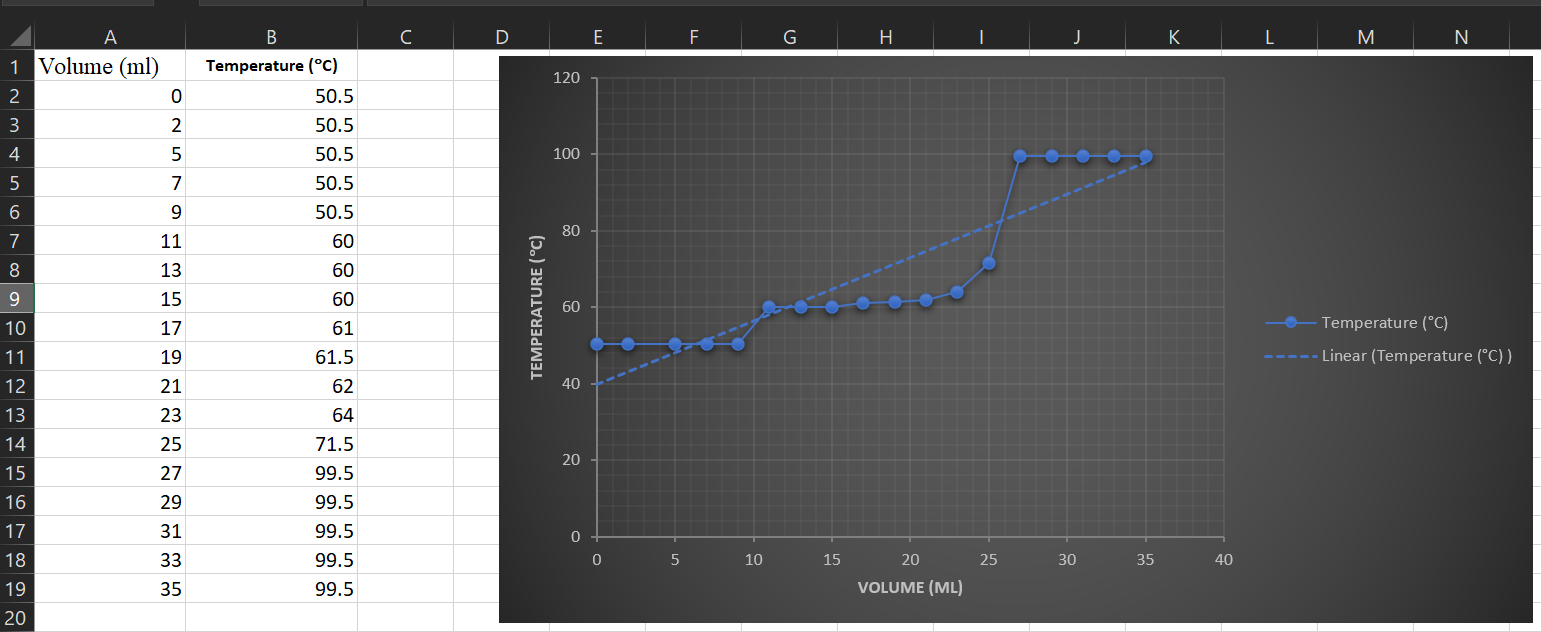

![PDF] Separation of Water-Acetone Mixture Using Suitable Entrainer (Simulation) | Semantic Scholar PDF] Separation of Water-Acetone Mixture Using Suitable Entrainer (Simulation) | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3b24cb3fdaba29b0182f3943cf35a512430d1196/29-Table3-2-1.png)



